Herbalife Things To Know Before You Get This
Herbalife Things To Know Before You Get This
Blog Article
Some Known Details About Herbalife
Table of ContentsNot known Factual Statements About Herbalife Herbalife Things To Know Before You Get ThisHerbalife Can Be Fun For EveryoneHerbalife Things To Know Before You Buy
Natural medications are typically marketed as food supplements, but a typical regulatory structure does not exist in different countries. As an outcome, information on scientific indications for their usage, efficacy, and safety are affected by the conventional experience offered in each area. A brief summary of the regulations in USA, copyright, and Europe is provided in this section, and might be made use of to direct the legal facets of the natural medication industry in various other nations.Dietary supplements do not require approval from the Food and Medication Management (FDA) before they are marketed (FDA 2010. herbalife pricing. Under DSHEA, natural medications, which are classified as nutritional supplements, are assumed secure, and the FDA does not have the authority to need them to be approved for security and efficiency before they go into the market, which is the instance for drugs
A dietary supplement supplier or supplier of a supplement with a "new dietary component," that is, an active ingredient that was not marketed in the United States before October 1994, might be called for to go with premarket testimonial for security data and other information. Additionally, all residential and foreign business that make package labels or hold nutritional supplements should adhere to the FDA's existing great production technique (GMP) regulations, which outline treatments for making sure the high quality of supplements intended for sale (FDA 2010; Gao 2010).
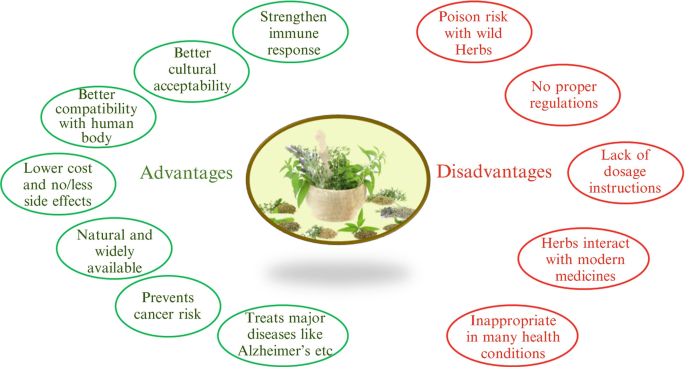
Herbalife Things To Know Before You Buy
In order to be granted a certificate, described details on the medical active ingredients, source, potency, nonmedicinal ingredients, and suggested use needs to be equipped. When an item has actually been given a license, it will birth the permit number and follow typical labeling requirements to guarantee that consumers can make enlightened choices.
On top of that, GMPs need to be employed to make sure item safety and top quality. https://padlet.com/isaacstokes2191/herbalife-y2vowrxqoiya8pjq. This requires that proper standards and practices regarding the manufacture, storage space, handling, and distribution of natural health items be satisfied. The GMPs are designed to be end result based, guaranteeing risk-free and premium items, while providing the flexibility to execute quality control systems appropriate to the line of product and service
In Europe, the European Directive 2004/24/EC released in 2004 by the European Parliament and by the Council of Europe provides the guidelines for using natural medicines (Calapai 2008 (herbalife products and pricing). The directive develops that organic medicines launched on the market demand permission by the nationwide regulatory authorities of each European nation and that these items need to have an acknowledged degree of security and efficiency (Calapai 2008
With regard to the production of these products and their high quality, products need to fulfill the very same demands as applications for an advertising consent. Details is based upon the availability of modern-day sciencebased public monographs in the European Pharmacopeia and their equivalents created by the pharmaceutical sector. The standards placed onward allow not just to define the high quality of items however additionally to remove damaging substances, debauchment, and contamination.
The Definitive Guide to Herbalife

The quality control of organic medications has a straight effect on their safety and security and effectiveness (Ernst, Schmidt, and Bigger 2005; Ribnicky et al. 2008. There is little data on the structure and top quality of the majority of organic medications not only due to absence of ample policies or government requirements but additionally due to an absence of appropriate or approved research methodology for evaluating typical medicines (WHO 2001; Kantor 2009. To isolate each energetic component from each herb would be tremendously time-consuming at a high price, making it not cost-efficient for makers (Richter 2003. One more trouble is that regardless of the appeal of herb dietary and herbal supplements, some organic products on the market are most likely to be of poor quality and suspect efficacy, also if the herb has actually been shown to have an effect in regulated research studies making use of high-quality item
Although herbs may well have unfavorable adverse effects, there are no collection "dosages," and herbdrug or herbherb communications are possible. A major theoretical benefit of botanicals over standard single-component medicines is the presence of numerous active substances that together can supply a potentiating effect that might not be achievable by any solitary compound.
:max_bytes(150000):strip_icc()/VWH_Illustration_10-Healing-Herbs-With-Medicinal-Benefits_Illustrator_Mira-Norian_Title_Final-47ce13013375448c9e8e7e8c21fb50f7.jpg)
An Unbiased View of Herbalife
The quality control of organic medicines has a direct influence on their safety and efficiency (Ernst, Schmidt, and Bigger 2005; Ribnicky et al. 2008. But, there is little data on the structure and top quality of a lot of natural medicines not just as a result of absence of appropriate policies or federal government needs but also due to an absence of appropriate or approved research methodology for examining conventional medications (WHO 2001; Kantor 2009. To separate each active component from each herb would be exceptionally time-consuming at a high price, making it not cost-efficient for suppliers (Richter 2003. One more problem is that regardless of the popularity of agricultural nutritional and natural supplements, some herbal items on the market are likely to be of poor quality and suspect effectiveness, even if the natural herb has actually been revealed to have an impact in regulated studies making use of top quality item
Although herbs might well have unwanted side results, there are no collection "doses," and herbdrug or herbherb communications are feasible. A significant theoretical benefit of botanicals over traditional single-component medications is the presence of several energetic substances that with each other can give a potentiating effect that may not be possible by any single substance.
Substances that are recognized by activity-guided fractionation should be checked in ideal pet versions to confirm in vivo task. Ideally, the make-up of the overall agricultural extract should be standardized and without any kind of possible hazards, and plants ought to be grown specifically for the manufacturing of agricultural extracts under controlled conditions and stem from a defined and consistent hereditary source with a browse around this web-site taxonomic document of the genus, varieties, and cultivar or various other additional identifiers.
Report this page